研究内容
細胞・組織バイオメカニクス
微小重力条件下における血管内皮細胞の形態的応答および血管新生
血管新生とは、既存血管から壁細胞が離脱することにより新たな血管が発芽し、この血管が伸長していく過程を指す。本研究ではBAECのコラーゲン・ゲル上培養を基にして、微小血管の血管壁を二次元的に再現した血管新生の観察用デバイスを作製し、微小重力負荷実験を行った。その結果、内皮細胞の増殖、形状の変化、細胞間接着の様子に有意な影響が見られた。本研究により、ガンの主原因ともされている血管新生の重力依存性を明らかにすることが可能となるため、宇宙空間への進出を想定したガン治療の新たな知見となることが期待される。
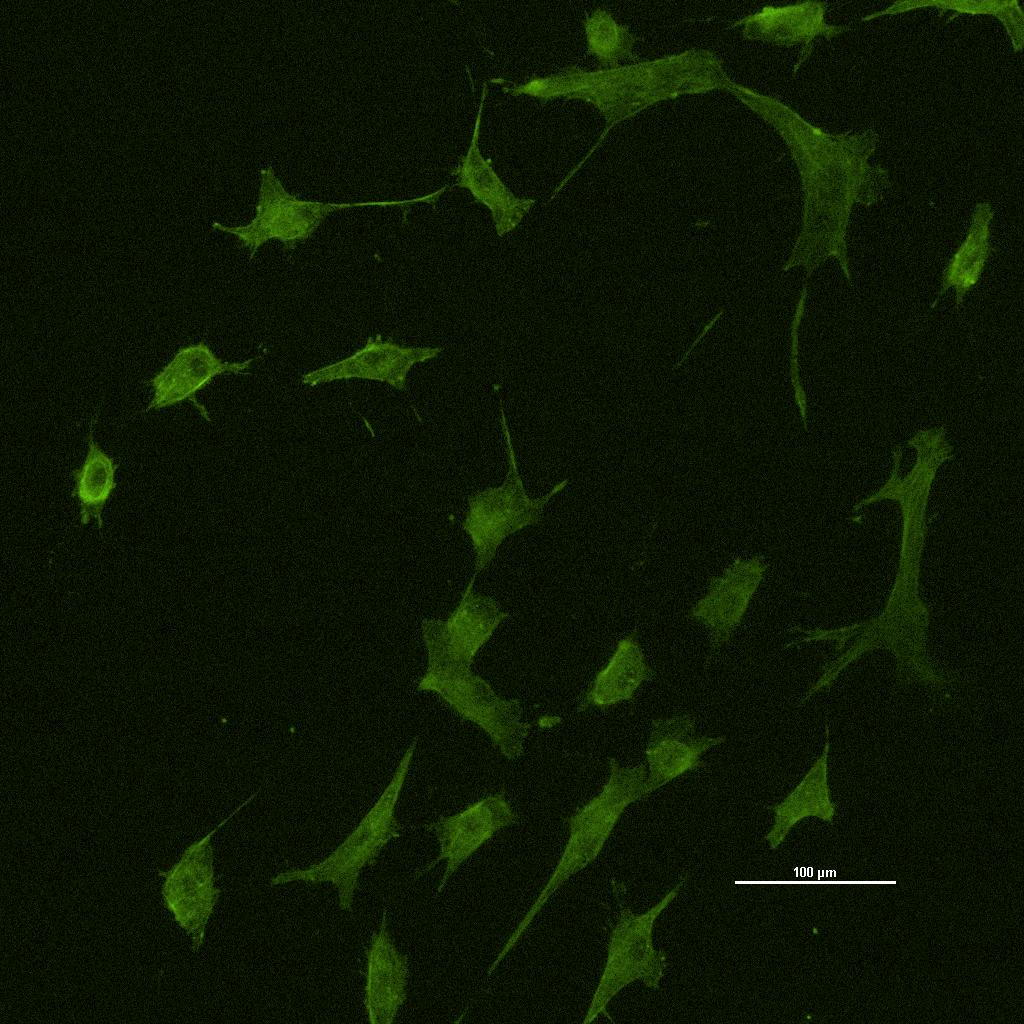

マイクロチャンネルを用いた細胞運動制御とがん細胞遊走への応用
この10年間で、生物学的実験から生成される大規模かつ複雑なデータセットの数が劇的に増加し、細胞、遺伝子、タンパク質、代謝物の存在量、マイクロバイオーム組成、集団全体の遺伝的変動などの分子変数が定量化されている。生物学研究において長年にわたり関心を集めているディープラーニングは、この複雑性に対処し、新たな視点を持ち、生命システムに関する新しい仮説を生み出すことを可能にする次世代分析を提供することを目的としている。本研究の目的は、Rac1によって解読される癌のリーダー細胞に対する新しい計算ベースの同定における深層学習である。このアプローチでは、免疫蛍光染色法に基づいてがんのリーダー細胞を同定し、抗体であるRac1のタイムラプスイメージングを観察する。本研究では、Rac1はもともと細胞運動を担うGTPaseであり、分子活動から形態変化への変換を表す応答関数を定量的に同定し、細胞内のラメラポディウムに多く存在することを明らかにした。
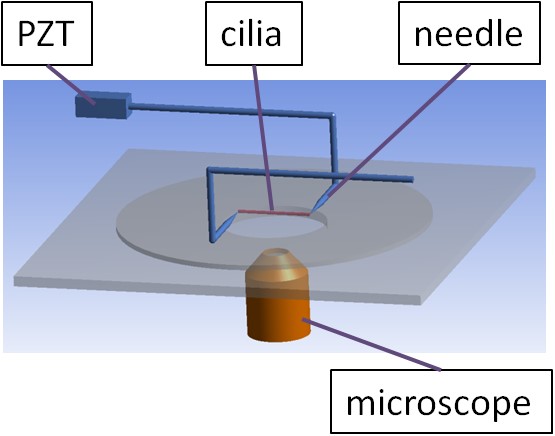
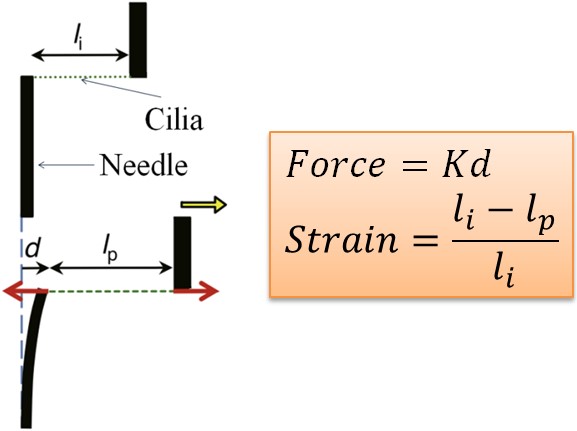
伸張試験による一次繊毛の力学的特性の測定
一次繊毛は、ほとんどの細胞の表面から突き出ている毛状の小器官である。そのため、一次繊毛の特性について関心が高まっている。 私たちは、自作の延伸装置を用いて、延伸試験による機械的特性の測定を試みている。ピエゾアクチュエータの動きは、ガラス棒を介して一次繊毛に伝達される。 ひずみは、一次繊毛の初期長さに対する全変形の割合として定義される。一次繊毛にかかる力は、変形量と針のバネ定数を掛け合わせたものである。 ひずみと力の関係から、ヤング率などの機械的特性を求めることを目的としている。
平滑筋細胞が発生する牽引力の測定に関する研究
細胞が受ける力学的環境の変化に応じて、細胞内構造が再編成されることが明らかにされている。特に、細胞牽引力は細胞と基質の相互作用に重要な役割を果たすと考えられており、細胞のリモデリングのプロセスに関与している可能性がある。微細加工技術を用いた細胞牽引力の推定に関する研究は数多く行われているが、牽引力の大きさや方向に対する機械的環境の影響、ひいては細胞内構造が牽引力にどのように寄与しているかについてはほとんど分かっていない。本研究では、これらの細胞骨格構造が平滑筋細胞の力学にどのように寄与しているのか、その基本的な側面を取り上げる。
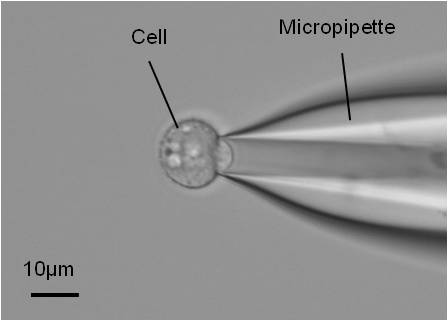
軟骨のメカノバイオロジー 機械的ストレスに応答する軟骨細胞の研究
軟骨の健康と軟骨組織工学への応用のためには、軟骨細胞のメカノトランスダクション(細胞が機械的信号を感知し応答する過程)を知ることが不可欠である。
私たちは、軟骨細胞の力学的特性を、マイクロピペット吸引法(細胞表面を小さなガラス管に吸引する方法)を用いて研究している。
この技術は、細胞のヤング率を測定することができ、細胞に機械的ストレスを与えるために利用することができる。
ウシ中手指節関節から軟骨細胞を分離するには、酵素を順次消化する方法がある。そこで、シリンジポンプで吸引圧を制御し、細胞のヤング率を測定する。
このプロジェクトは、ロンドン大学クイーン・メアリー校のマーティン・ナイト博士、ダン・ベイダー教授と共同で行っている。
バイオMEMS
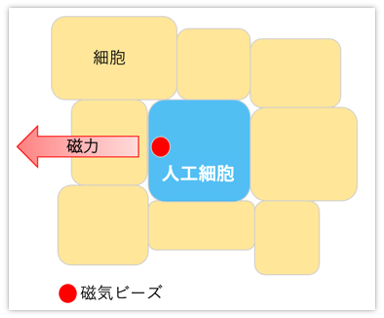
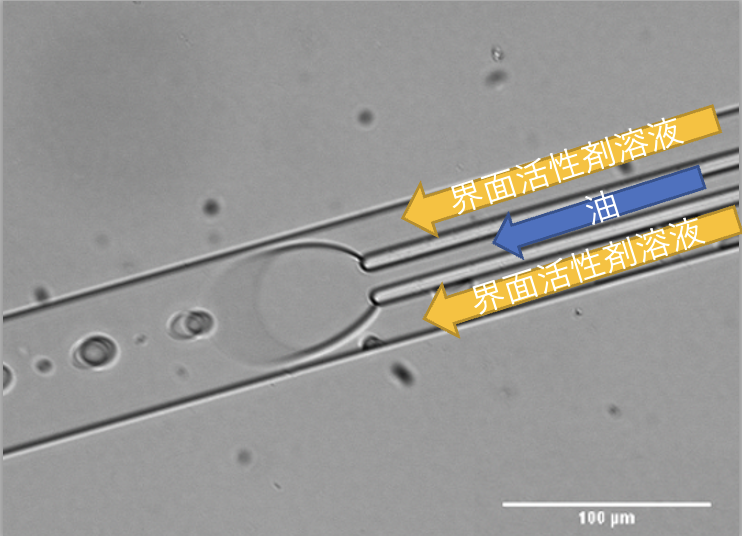
磁気ビーズを封入した人工細胞および作製用マイクロ流路デバイス開発
全ての多細胞生物は、細胞同士が互いに接着しあって様々な組織や臓器を構成している。細胞同士が互いに接着する場合、細胞間には力が発生している。この細胞間力は細胞集団の機能や生理において重要な役割を果たすことが示唆されている。
細胞間力は細胞生理を理解する上で非常に重要であるが、計測・制御の困難さから十分な研究が行われてきていない。
そこで我々の研究グループでは、生細胞内に配置された磁気ビーズ内包人工細胞を外部磁場により駆動させることで細胞間力を任意に計測・制御できる実験系を考案している。
また、人工細胞の大きさと磁気ビーズの封入量の安定化のためのマイクロ流路デバイスの開発も行っている。
周期的伸張に対する筋線維芽細胞の力学的応答
創傷治癒は、炎症期、成長期、成熟期の3つの時期に分けられる。成長期には筋線維芽細胞が増殖し、一定の方向に整列する。
筋線維芽細胞が一定方向に揃えば傷は早期に治癒し、揃わなければ傷の治癒は遅れる。この仮説を明らかにするため、筋線維芽細胞の伸縮実験を、伸縮速度1Hz、伸縮量20%で行った。
伸展後、顕微鏡で筋原線維の並びを観察する。同時に、細胞骨格を観察するために、アクチンフィラメントの蛍光染色とビンキュリンの免疫蛍光染色を行う。
計算バイオメカニクス
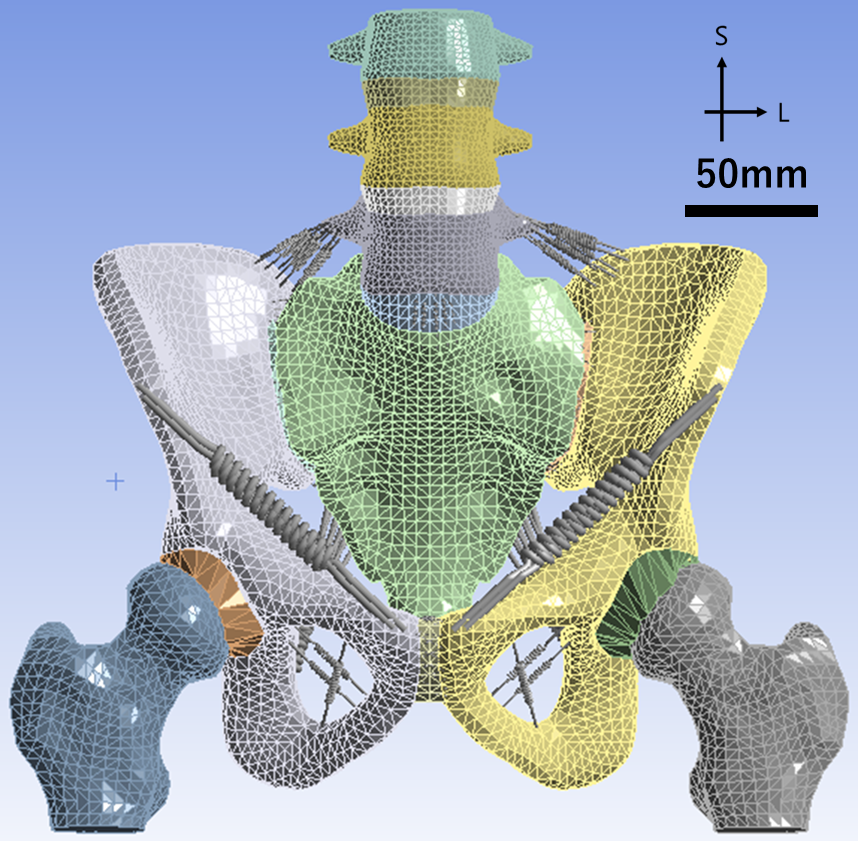
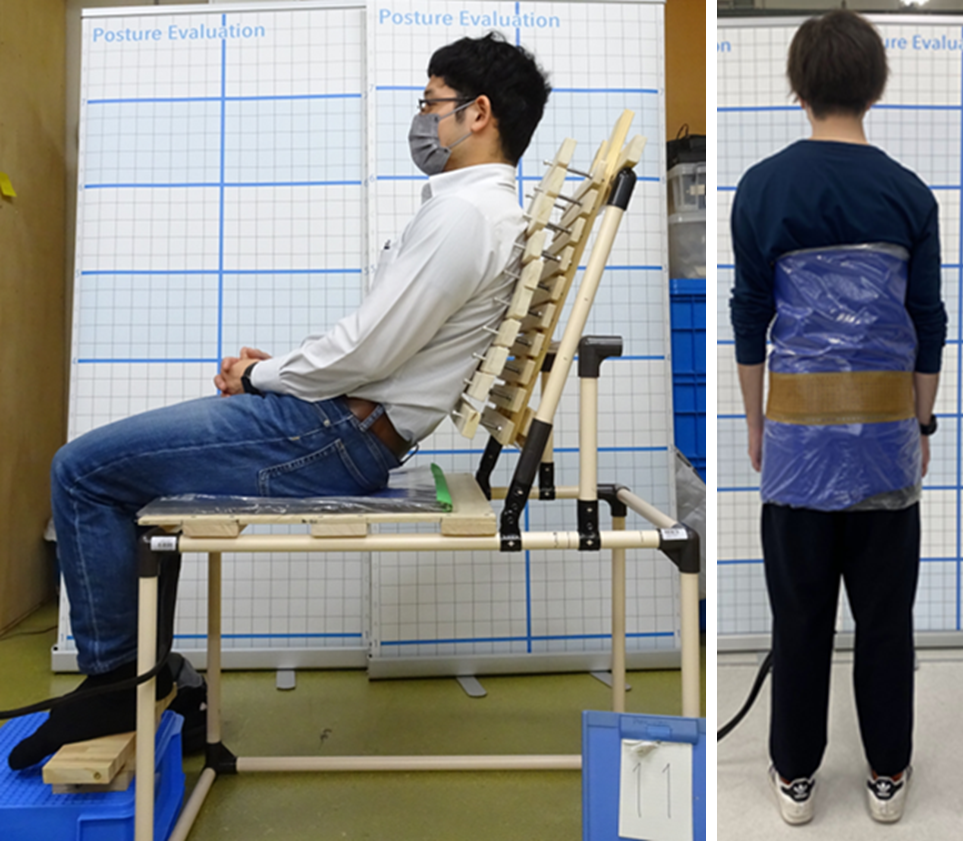
ヒト骨盤内仙腸関節のバイオメカニクス
仙腸関節は仙骨と腸骨の間に存在する関節で、通常は強固な靱帯により固定されていますが急な動作や繰り返しの負荷によって関節にズレが生じ、疼痛を引き起こすことがあります(仙腸関節疾患)。この疾患はレントゲンやCTなどの画像検査を用いて体外から評価することが難しいため、有限要素法(FEM)を活用して骨盤内の応力状態を解析・評価しています。
仙腸関節の機能理解や疾患発生の機序解明、疼痛が軽減する姿勢の調査、効果的な治療法の提案を目的としています。有限要素モデルの作成や解析入力値の測定も行っています。
植物バイオメカニクス
風洞実験を用いた力学刺激と植物の成長に関する検討
マイクロ波刺激や微小重力環境で植物の育成を行うとそれぞれの刺激によって様々な反応の違いを示す。そこで、本研究では植物に風を当てた時に成長にどのような影響をあたえるのかを目的とし解析を行った。風洞でシロイヌナズナに2~3週間風を当て続け、その変化をデジタル画像相関法で解析を行った。これにより、植物内に発生する応力と植物の成長速度の関係性を明らかにした。さらに、相対成長率を調べることで、風による成長の変化は生理学的要因によるものか、形態学的要因によるものかを調べることを可能とした。


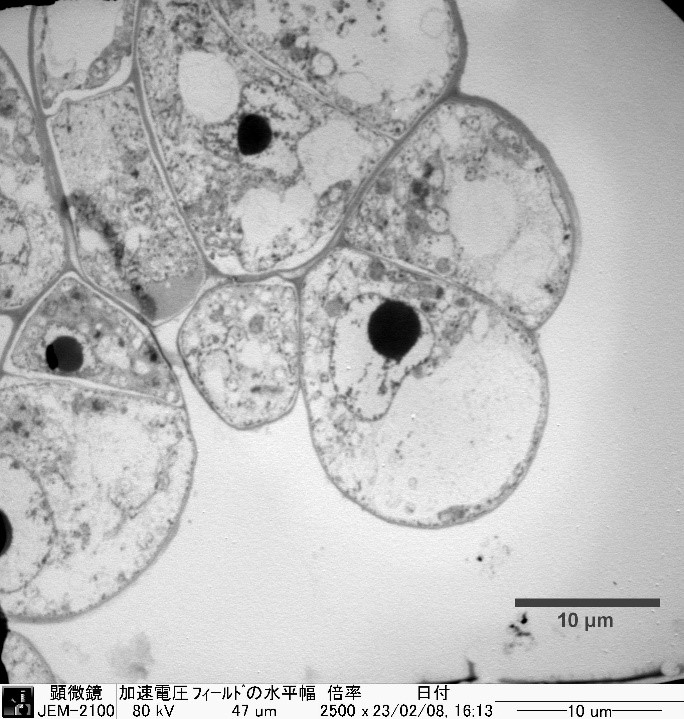
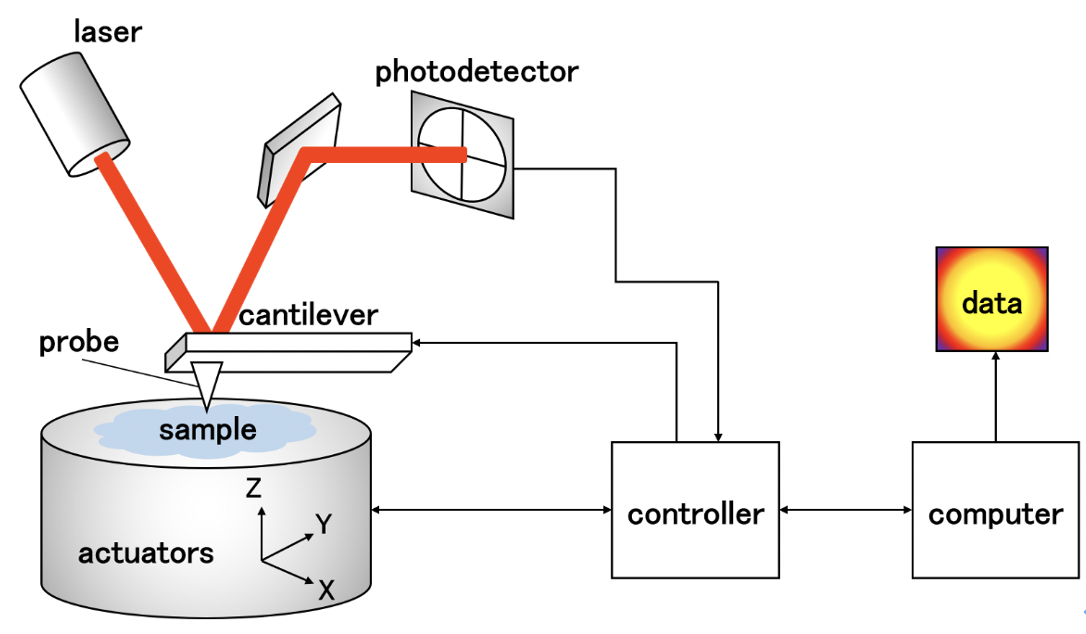
シロイヌナズナ植物細胞の力学特性計測
一方向から風を受け続けた樹木が湾曲するように植物の成長は外部刺激の影響を受ける。本研究では外部刺激に対する植物の応答を知ることを目指し、植物細胞の力学特性を計測する。植物細胞の力学特性に大きな影響を及ぼす植物細胞壁は層構造を有している。そこで、透過型電子顕微鏡(TEM)で細胞壁各層の厚さを計測し、層構造を反映した3Dモデルを作成する。そのモデルと原子間力顕微鏡(AFM)で得た押し込み深さ毎の力学応答のデータを組み合わせることで各層の力学特性を解析する。